Other grades of this product :
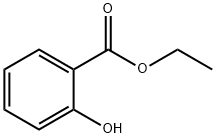
| Ethyl 2-hydroxybenzoate Basic information |
| Ethyl 2-hydroxybenzoate Chemical Properties |
| Hazard Codes | Xn,Xi | | Risk Statements | 22-36/38-52/53 | | Safety Statements | 26-36 | | WGK Germany | 1 | | RTECS | VO3000000 | | TSCA | Yes | | HS Code | 29182390 | | Toxicity | LD50 orally in Rabbit: 1320 mg/kg LD50 dermal Rabbit > 5000 mg/kg |
| Ethyl 2-hydroxybenzoate Usage And Synthesis |
| Description | Ethyl 2-hydroxdybenzoate is also known as Ethyl salicylate, which a kind of ester formed through the condensation between salicylic acid and ethanol. It can be used as a perfumery, artificial essence flavoring agent and used in cosmetics. It can also be used as analgesics, anti-inflammatory and antipyretic agents.
| | Chemical Properties | clear colourless to pale yellow liquid | | Chemical Properties | Ethyl salicylate is the ester formed by the condensation of salicylic acid and ethanol. It is a clear liquid that is sparingly soluble in water, but soluble in alcohol and ether. It has a pleasant odor resembling winter green and is used in perfumery and artificial flavors. | | Chemical Properties | Ethyl salicylate has a characteristic aromatic odor similar to wintergreen. It tends to darken on exposure to light and air. | | Occurrence | Reported found in raspberry, tomato, currants, rum, whiskey, red wine, plum brandy, Cape gooseberry, Feijoa
fruit, papaya and scotch. | | Uses | manufacture of artificial perfumes. | | Uses | Ethyl salicylate is a non-steroidal analgesic, anti-inflammatory and antirrheumatic drug. Its complexation with β-cyclodextrin was studied using reversed-phase liquid chromatography. | | Uses | Ethyl salicylate may be used as an analytical standard for the determination of the analyte in biological fluids and topical formulation by chromatography-based techniques. | | Preparation | By esterification of salicylic acid with ethyl alcohol and concentrated H2SO4 at 100°C in the presence of aluminum sulfate; by heating to the boil an alkaline solution of salicylic acid and ethyl p-toluenesulfonate. | | Production Methods | Ethyl salicylate is found naturally in currants and strawberries
. It is manufactured commercially by the esterification
of salicylic acid with ethyl alcohol. | | Taste threshold values | Taste characteristics at 10 ppm: sweet, wintergreen, spicy and anisic. | | General Description | Ethyl salicylate was hydrolysed to salicylic acid by the use of an extractive electrospray ionisation (EESI) device. | | Flammability and Explosibility | Notclassified | | References | Sheu, Yaw-Wen, and Chein-Hsiun Tu. "Densities and viscosities of binary mixtures of ethyl acetoacetate, ethyl isovalerate, methyl benzoate, benzyl acetate, ethyl salicylate, and benzyl propionate with ethanol at T=(288.15, 298.15, 308.15, and 318.15) K." Journal of Chemical & Engineering Data 51.2 (2006): 545-553.
Filippa, Mauricio, Matías I. Sancho, and Estela Gasull. "Encapsulation of methyl and ethyl salicylates by β-cyclodextrin: HPLC, UV–vis and molecular modeling studies." Journal of pharmaceutical and biomedical analysis 48.3 (2008): 969-973.
|
| Ethyl 2-hydroxybenzoate Preparation Products And Raw materials |
|