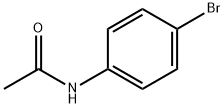
Other grades of this product :
| 4'-Bromoacetanilide Chemical Properties |
| Melting point | 165-169 °C(lit.) | | Boiling point | 353.4°C (estimate) | | density | 1.72 | | refractive index | 1.6120 (estimate) | | storage temp. | Store below +30°C. | | solubility | alcohol: moderately soluble | | pka | 14.25±0.70(Predicted) | | form | Crystalline Powder or Crystals | | color | White to beige or light gray | | Water Solubility | Insoluble | | Merck | 14,1399 | | BRN | 2208091 | | Stability: | Stable. Incompatible with strong oxidizing agents. | | CAS DataBase Reference | 103-88-8(CAS DataBase Reference) | | NIST Chemistry Reference | Acetamide, N-(4-bromophenyl)-(103-88-8) | | EPA Substance Registry System | Acetamide, N-(4-bromophenyl)- (103-88-8) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | RIDADR | 2811 | | WGK Germany | 3 | | RTECS | AD9625000 | | Hazard Note | Irritant | | TSCA | Yes | | HazardClass | 6.1(b) | | PackingGroup | III | | HS Code | 29242998 |
| 4'-Bromoacetanilide Usage And Synthesis |
| Description | 4'-Bromoacetanilide is a Pale yellow crystals or powdery crystals.Melting point 168℃, relative density 1.717.Soluble in benzene, chloroform, ethyl acetate, slightly soluble in alcohol, slightly soluble in hot water, insoluble in cold water.
It can be used as reparation of 4'-Bromoacetanilide from Acetanilide.Principle: Aromatic compounds can be conveniently brominated by the use of brominating agent, which is normally a solution of bromine in acetic acid. Bromination of activated aromatic compounds usually give 2, 4, 6-tribromo derivatives while moderately activating group like anilide give preferably the para bromo product. | | Uses | 4'-Bromoacetanilide can be used as an intermediate in organic synthesis. | | Chemical Properties | 4'-Bromoacetanilide is a white to light beige crystalline solid. | | Uses | 4'-Bromoacetanilide is used as an internal standard for phenylurea and the respective metabolic products in oysters. It has also been used as a reagent in the production of a new ligand to form a coordination compex between gadolinium(III) and T=titanium(IV) oxide. It was used in the synthesis of new ligand for anchoring Gd(III) chelates onto TiO2 surface. | | Uses | 4-Bromoacetanilide was used as internal standard in determination of several phenylurea and triazine herbicides and their transformation products in oyster. It was used in the synthesis of new ligand for anchoring Gd(III) chelates onto TiO2 surface. | | Preparation | Preparation of 4'-Bromoacetanilide from Acetanilide.
Principle: Aromatic compounds can be conveniently brominated by the use of brominating agent, which is normally a solution of bromine in acetic acid. Bromination of activated aromatic compounds usually give 2, 4, 6-tribromo derivatives while moderately activating group like anilide give preferably the para bromo product.
Reaction:
Procedure: Dissolve 0.5 g acetanilide in 0.6 ml glacial acetic acid and add 2.5 ml bromine solution in acetic acid (25% w/v). Shake the mixture for 1 h. After 1 h, pour the mixture on to crushed ice (20 g) with stirring. Filter the separated product and wash with cold water. Dry the product, record the practical yield and re-crystallize it.
Re-crystallization: Dissolve the crude product in minimum amount of ethyl alcohol in a beaker by heating on a water bath. Filter the hot solution and cool the filtrate. The crystals of the product separate out. Filter, dry and record the melting point and TLC (using toluene as a solvent). | | Synthesis Reference(s) | Canadian Journal of Chemistry, 50, p. 1233, 1972 DOI: 10.1139/v72-193Synthetic Communications, 23, p. 779, 1993 DOI: 10.1080/00397919308009839 | | Purification Methods | Crystallise the anilide from aqueous MeOH or EtOH. Purify it by zone refining. [Beilstein 12 IV 1504.] |
| 4'-Bromoacetanilide Preparation Products And Raw materials |
|