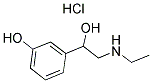
| Etilefrine hydrochloride Basic information |
| Product Name: | Etilefrine hydrochloride |
| Synonyms: | alpha-((ethylamino)methyl)-m-hydroxybenzylalcoholhydrochloride;3-[2-(ETHYLAMINO)-1-HYDROXYETHYL]PHENOL HYDROCHLORIDE;2-ETHYLAMINO-1-(3-HYDROXYPHENYL)ETHANOL HYDROCHLORIDE;LABOTEST-BB LT00244796;Etilefrina;ETILEFRIN HYDROCHLORIDE 98+%;(+-)-Ethylphenylephrine hydrochloride;dl-n-ethylnorphenylephrine hydrochloride |
| CAS: | 943-17-9 |
| MF: | C10H16ClNO2 |
| MW: | 217.69 |
| EINECS: | 213-398-8 |
| Product Categories: | |
| Mol File: | 943-17-9.mol |
| Etilefrine hydrochloride Chemical Properties |
| Melting point | 122 °C |
| solubility | Freely soluble in water, soluble in ethanol (96 per cent), practically insoluble in methylene chloride |
| form | neat |
| Safety Information |
| RIDADR | UN 2811 6.1 / PGIII |
| RTECS | DO5618000 |
| HS Code | 2922290090 |
| MSDS Information |
| Etilefrine hydrochloride Usage And Synthesis |
| Chemical Properties | White or almost white, crystalline powder or colourless crystals. |
| Originator | Circupon,Troponwerke,W. Germany |
| Uses | Adrenoceptor agonist. |
| Manufacturing Process | 30 parts of 1-(3'-hydroxyphenyl)-2-(N-benzylaminomethyl)-ethan-1-one are mixed with 100 parts of pyridine and 30 parts of pivalic anhydride and dissolved while warming. After heating for 1 hour under reflux, the acylation is complete. After concentrating the reaction solution, the product is precipitated from acetone/ether. Yield: 96.4% of 1-(3'-pivaloyloxyphenyl)-2- (N-benzylaminomethyl)-ethan-1-one.3 parts of palladium/charcoal (10% strength) are prehydrogenated in water, thereafter 10 parts of 1-(3'-pivaloyloxyphenyl)-2-(N-benzylaminoethyl)-ethan- 1-one, dissolved in a 10-fold amount of water, are added dropwise at room temperature and hydrogenation is carried out until 1 mol of hydrogen has been taken up. After filtering off the catalyst, a further 3 parts of palladium/charcoal are added and hydrogenation is carried out until a further mol of hydrogen has been taken up. The catalyst is separated off and after removal of the solvent the hydrogenation product is reprecipitated from acetone/petroleum ether and from methanol/ether until it is pure according to thin layer chromatography. Yield: 38.8% of 1-(3'-pivaloyloxyphenyl)-2- ethylaminoethanol-1 hydroxide, melting point 208°C to 209°C. |
| Therapeutic Function | Tranquilizer |
| Etilefrine hydrochloride Preparation Products And Raw materials |
| Raw materials | Hydrogen-->TRIMETHYLACETIC ANHYDRIDE |
Please leave a message for us or use the following ways to contact us, we will reply to you as soon as possible, and provide you with the most sincere service, thank you.