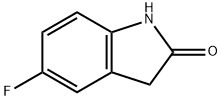
| 5-Fluoro-2-oxindole Basic information |
| Product Name: | 5-Fluoro-2-oxindole |
| Synonyms: | 5-Fluoroindole-2-One;5-Fluoro-1,3-dihydro-indol-2-one;5-FLUORO-2-OXINDOLE;5-FLUORO-2-OXYINDOLE;5-FLUORO-1,3-DIHYDRO-2H-INDOL-2-ONE;5-Fluorooxindole 98%;5-Fluorooxindole98%;5-FLUROOXINDOLE |
| CAS: | 56341-41-4 |
| MF: | C8H6FNO |
| MW: | 151.14 |
| EINECS: | 638-803-9 |
| Product Categories: | Fluorinated heterocyclic series;Indoles and derivatives;Indole/indoline/oxindole;Indole and Indoline;Heterocycles;Aromatics Compounds;Halides;Pyrroles & Indoles;ketone;Sunitinib;Intermediate of sunitinib malate;Aromatics;Pyrroles & Indoles;Boronic Acid;INDOLE;blocks;FluoroCompounds;IndolesOxindoles;Heterocyclic Compounds;Building Blocks;Heterocyclic Building Blocks;Indoles;Intermediates;56341-41-4 |
| Mol File: | 56341-41-4.mol |
| 5-Fluoro-2-oxindole Chemical Properties |
| Melting point | 143-147 °C (lit.) |
| Boiling point | 307.2±42.0 °C(Predicted) |
| density | 1.311±0.06 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| pka | 12.99±0.20(Predicted) |
| form | Powder or Crystalline Powder |
| color | White to pale brown |
| InChIKey | DDIIYGHHUMKDGI-UHFFFAOYSA-N |
| CAS DataBase Reference | 56341-41-4(CAS DataBase Reference) |
| Safety Information |
| Hazard Codes | Xi,C |
| Risk Statements | 36/37/38-34 |
| Safety Statements | 22-24/25-45-36/37/39-27-26 |
| WGK Germany | 3 |
| Hazard Note | Irritant/Keep Cold |
| HazardClass | IRRITANT, KEEP COLD |
| HS Code | 29339900 |
| MSDS Information |
| Provider | Language |
|---|---|
| SigmaAldrich | English |
| 5-Fluoro-2-oxindole Usage And Synthesis |
| Chemical Properties | Light Yellow Crystalline |
| Uses | 5-Fluoro-2-oxindole (cas# 56341-41-4) is a compound useful in organic synthesis. |
| Application | 5-Fluoro-2-oxindole is a ketone organic compound that can be used as an intermediate in the preparation of sunitinib. Sunitinib is a novel multi-targeted oral drug for the treatment of tumors. |
| Preparation | synthesis of 5-fluoro-2-oxindole: 4-fluoroaniline was used as the starting material, reacted with chloral hydrate and hydroxylamine hydrochloride to generate 4-fluoroisonitrosoacetanilide, and then cyclized under the action of concentrated sulfuric acid to form the compound 5-fluoroisatin was finally obtained by wolff-kishner-huang minlon reduction to obtain 5-fluoro-2-oxindole with a total yield of 66%. |
| 5-Fluoro-2-oxindole Preparation Products And Raw materials |
Please leave a message for us or use the following ways to contact us, we will reply to you as soon as possible, and provide you with the most sincere service, thank you.