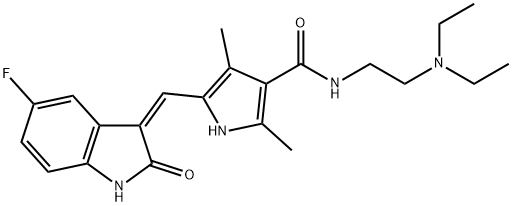
| Description | Sunitinib is an inhibitor of multiple receptor tyrosine kinases (RTKs) involved
in tumor proliferation and angiogenesis, including platelet-derived growth factor
receptors (PDGFR), vascular endothelial growth factor receptors (VEGFR), and
stem cell factor receptor (KIT). It was launched as an oral treatment for
gastrointestinal stromal tumors (GIST) and advanced renal-cell carcinoma (RCC).
In vitro, sunitib inhibits VEGFR2, PDGFRα, PDGFRβ, KIT, and FLT3 receptors
with IC50 values in the 4–14nM range, and the ligand-dependent autophosphorylation
of VEGFR2 and PDGFRb with IC50s of approximately 10 nM.
In addition, it inhibits the growth of tumor cells expressing dysregulated target RTKs in vitro and inhibits PDGFRb- and VEGFR2-dependent tumor angiogenesis
in vivo. Sunitinib exhibits broad and potent antitumor activity, causing regression
in murine models of human epidermal (A431), colon (Colo205 and HT-29), lung
(NCI-H226 and H460), breast (MDA-MB-435), prostate (PC3-3M-luc), and renal
(786-O) cancers, and suppressing or delaying the growth of many others,
including the C6 rat and SF763 T human glioma xenografts and B16 melanoma
lung cancer. |
| Chemical Properties | Yellow Solid |
| Originator | Sugen (US) |
| Uses | A multi-kinase inhibitor targeting several receptor tyrosine kinases (RTK). Antineoplastic |
| Uses | Sunitinib Malate (Sutent, SU11248) is a multi-targeted RTK inhibitor targeting VEGFR2 (Flk-1) and PDGFRβ with IC50 of 80 nM and 2 nM, and also inhibits c-Kit. |
| Uses | Sunitinib Malate is a multi-targeted RTK inhibitor targeting VEGFR2 (Flk-1) and PDGFRβ with IC50 of 80 nM and 2 nM, and also inhibits c-Kit. |
| Uses | Labelled Sunitinib (S820000), a multi-kinase inhibitor targeting several receptor tyrosine kinases (RTK). |
| Uses | Labelled Sunitinib, a multi-kinase inhibitor targeting several receptor tyrosine kinases (RTK). |
| Brand name | (Pfizer). |
| Clinical Use | Tyrosine kinase inhibitor:
Treatment of metastatic renal cell carcinoma
(MRCC), gastrointestinal stromal tumours (GIST)
and pancreatic neuroendocrine tumours (pNET) |
| Drug interactions | Potentially hazardous interactions with other drugs
Antipsychotics: avoid with clozapine (increased risk
of agranulocytosis).
Antivirals: avoid concomitant use with boceprevir.
Avoid concomitant use with other inhibitors or
inducers of CYP3A4. Dose alterations may be
required. |
| Metabolism | Metabolised mainly via the cytochrome P450 isoenzyme
CYP3A4 to its primary active metabolite, which itself is
then further metabolised via CYP3A4.
Elimination is primarily via faeces. In a human mass
balance study of [14C]sunitinib, 61% of the dose was
eliminated in faeces and 16% by the renal route. |
| references | [1]hui ep1, lui vw, wong cs, ma bb, lau cp, cheung cs, ho k, cheng sh, ng mh, chan at. preclinical evaluation of sunitinib as single agent or in combination with chemotherapy in nasopharyngeal carcinoma. invest new drugs. 2011 dec;29(6):1123-31. |